AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Molecular orbital theory diagram1/15/2024  Compare the bond order to that seen in the Lewis structure (remember that an electron in an antibonding orbital cancels the stabilization due to bonding of an electron in a bonding orbital). Bonding between atoms occurs when they come into close enough proximity for their orbitals to overlap. Construct a qualitative molecular orbital diagram for chlorine, Cl 2. To further demonstrate the consistency of the. The work also gives a clear definition of the terms bonding, non-bonding and antibonding molecular orbitals. Molecular Orbital Theory - The goal of molecular orbital theory is to describe molecules in a similar way to how we describe atoms, that is, in terms of orbitals, orbital diagrams, and electron configurations. The orbital correlation diagram in predicts the same thing-two electrons fill a single bonding molecular orbital. Walsh's original diagrams seem to be based on an unusual type of delocalized molecular orbital. If these distinctions are not made, one may emerge with a diagram which is a mixture of several different but related diagrams. Molecular orbital diagram is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the. 
The atomic orbitals combine to produce the following molecular orbital diagram: Here the 2p g orbital is occupied by two electrons to give a total bond order of three. The general method is to reduce the self-consistent field molecular orbital theory to the independent electron theory by systematic approximations and by appeal to physical arguments.īroadly speaking, the results support Walsh's method of constructing his diagrams but they also show that it is important to distinguish clearly between ionization energies and binding energies and also between the different types of delocalized molecular orbital. Pauling with others, developed Resonance Theory, which provided the rationale to an all-embracing orbital theory. This interpretation is discussed in the context of the Walsh diagram for the water molecule. 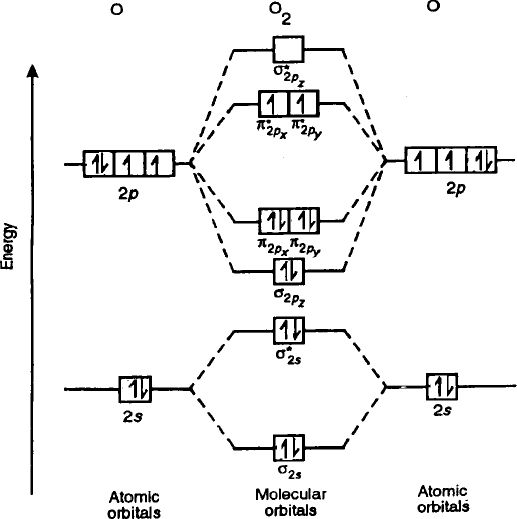
It is suggested that the “one electron energy” or “binding energy” of the independent electron molecular orbital theory is the change in the ionization energy of the molecular orbital on molecule formation. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
